One column fits all was title of my poster presented at HPLC 2010 in Boston that described the preparation and characterization of hypercrosslinked monolithic stationary phases and their application in several chromatographic modes. We summarized poster’s results and submitted them as a paper in Journal of Chromatography A.
Hypercrosslinking modification is not a new technique. Several decades ago, Davankov prepared large surface area polymers using this approach. However, this approach is a new in the field of monolithic stationary phases. It allows us to prepare materials with both large flow-through pores and small pores on the surface of monolithic scaffold.
On the beginning of this year, we published short letter describing the application of hypercrosslinking modification of monolithic stationary phases in Analytical Chemistry.
So what exactly is hypercrosslinking modification and what is new in the newest paper?
Hypercrosslinking modification
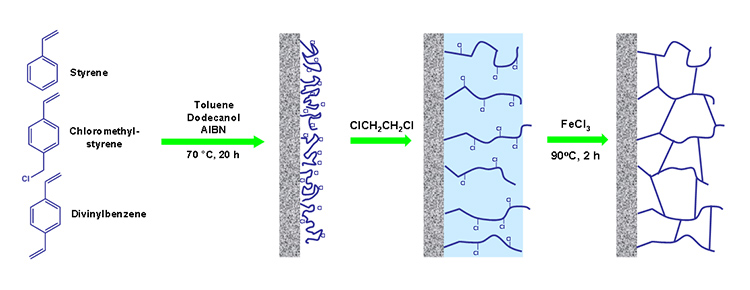
The difference in reactivity ratios for monomers lead to porous polymer which allow hypercrosslinking. The divinylbenzene polymerizes faster then monovinyl styrene and vinylbenzyl chloride. Thus, remaining monomer mixture becomes significantly richer in the monovinyl monomers as the polymerization reaction approaches completion and affords only slightly crosslinked chains attached to the surface of polymerized monolith. After solvation with a thermodynamically good solvent, this layer can be crosslinked via the Friedel-Crafts reaction. The polymer chains become fixed in their solvated state during the reaction thus forming pores that persist even after the solvent is removed.
Following scheme shows each step of hypercrosslinking modification.
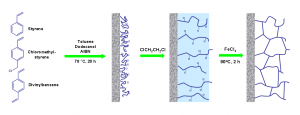
As I said, hypercrosslinking allows to prepare monolithic materials with both large and small pores. With this approach we can overcome the usual problem of organic polymer monolithic stationary phases – their weak separation power in terms of separation of small molecules (although, I have to say that other directions, such as short time of polymerization, provide also monoliths suitable for these kind of separations).
The presence of small pores in the monolithic material enhances significantly its surface area. Using this approach we were able to prepare monolithic materials with surface area higher than 600 m2/g.
Polymerization mixture composition
In the first paper about hypercrosslinked monoliths, we have shown that the final properties of hypercrosslinked monolithic stationary phases strongly depends on the composition of the polymerization mixture. In the new one, we studied the influence of the polymerization mixture composition more deeply using mixture design approach. We systematically varied the composition of the polymerization mixture and compared the resulting properties (efficiency and porosity) with concentration of individual compounds in the mixture.
As expected, we found out that extend of hypercrosslinking depends on the percentage of divinylbenzene in the monomer mixture, which controls the number and length of hypercrosslinkable loose chains, and the ratio of functional monomer vinylbenzyl chloride and inert styrene that affects the frequency of reactive sites along these loose chains.
Time and temperature of hypercrosslinking modification
We tested influence of time and temperature of hypercrosslinking modification. There is no change in column mesopore porosity or efficiency after 2 hours of modification, which agrees with previously published data.
In terms of temperature, the concentration of mesopores inside the hypercrosslinked monoliths as well as an efficiency of the column increases with increase in modification temperature. However, there is no increase in these properties with temperatures higher than 90°C. Thus, the best columns are prepared with hypercrosslinking modification for 2 h at 90°C.
Mobile phase composition
Since the beginning of this project, we were facing the problem with peak tailing, especially for more retained compounds. Then I found in the literature (and here), that peak tailing is quite common problem for styrene-divinylbenzene type of the stationary phases.
This issue can be solved by adding thermodynamically good solvent in the mobile, such as tetrahydofuran (THF). With higher concentration of THF in the mobile phase the peak shape improves and retention decreases. The optimum composition of the mobile phase was found to be 20% water, 20% THF and 60% acetonitrile.
Effect of temperature
High temperature liquid chromatography is experiencing kind of come back during last couple of months/years. We tested influence of higher analysis temperature on the separation power of hypercrosslinked monoliths. With higher temperature the separation time of six alkylbenzenes (benzene through amylbenzene) decreases from 8 minutes at 20 °C to less then 4 minutes at 80°C.
Analysis temperature has also positive effect on the efficiency of the (hypercrosslinked) columns. The minimum of van Deemter curve for benzene at optimized ternary mobile phase decreases from 28 μm at 20 °C to 16 μm at 80 °C.
Sample loading
Couple of years ago, I have studied effect of injected volume and mass on the efficiency of the monolithic columns (silica-based Merck). At that time, I did not find any significant decrease in column performance with higher injected volume or mass. It was part of another study and we never published those data but since then I always thought something in the sense that it is very difficult to overload monolithic column.
I have to say I would never believe that the injected concentration might play significant role in capillary format organic polymer monoliths. It did.
With decrease in injected amount of benzene the column efficiency increased significantly. Keeping all conditions constant and changing only the amount of benzene, the efficiency increases twice with decrease in the mass from 17.5 to 0.14 pg.
Over 80 000 theoretical plates/m
The van Deemter curve for benzene (0.14 pg) measured at 80°C using optimized ternary mobile phase (20% water, 20% THF and 60% acetonitrile) shows the minimum at 12 μm. This value corresponds to the column efficiency 83 200 theoretical plates/m and represents the highest column efficiency found for a organic polymer monolithic column used in isocratic mode.
Optimized separations
Taking into account all previously mentioned steps of optimization (polymerization mixture composition, hypercrosslinking time & temperature, mobile phase composition, and analysis temperature) we are able to show very fast and efficient separation of small molecules.
Our testing mixture contains small alkylbenzenes (benzene, toluene, ethylbenzene, propylbenzene, butyl benzene and amylbenzene) and we are able to separate them in less then 2 min at elevated temperature. Moreover, this type of the columns proved to be successful also in separation of peptides in gradient mode.
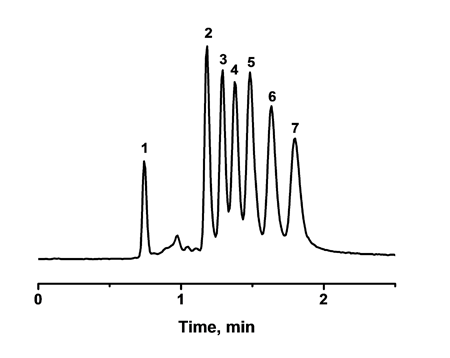
Size-exclusion chromatography
Last but not least, the advantage of hypercrosslinked monolithic stationary phases is significant amount of small pores in their pore size distribution. Thus, they can be used for size-exclusion type of separations. Indeed, we were able to separate four polystyrene standards and toluene in less then 10 minutes using two hypercrosslinked columns connected with zero-volume union (total length 670 mm).
It is the first demonstration of the use of monolithic column in size-exclusion chromatography of polymers using an organic solvent as the mobile phase.
And I have to say – finally! – because I already tried to prepare such a column couple of years ago.
Uff, that is it. Long post. If you are interested in hypercrosslinked monolithic stationary phases you might either read the paper published in Journal of Chromatography A or download the poster I presented at HPLC 2010 in Boston (pdf, 5.7 MB).
What do you think about hypercrosslinked monolithic stationary phases? Do you think that this 2nd generation of organic polymer monoliths can catch up current highly efficient columns with superficially porous particles?
If you like this post or maybe even whole website you might consider to subscribe to RSS channel or my newsletter.
3 replies on “One column fits all – Hypercrosslinked monoliths”
[…] Then, there is no room for anything else than Stationary phases section. I would like to see talk of my boss Pavel Jandera in Multidimensional separations, but I have to be ready for my talk in my section (I would really appreciate if you come and see talk about hypercrosslinked monoliths). […]
[…] their separation power for small molecules. It is possible to increase their surface area either by hypercrosslinking modification or with the combination of non-polar and polar monomers. Another possibility is to prepare […]
[…] four of us, Tom and Nick as NMR guys and Stuart and me responsible for chromatography. We have used hypercrosslinked monolithic columns which already proved to be suitable for fast separation of small […]